Yusuf Dilgin seminar
Ilaria Fratoddi is pleased to invite to the seminar:
Electrochemical sensors and biosensors using chemically modified disposable electrodes
di Prof. Dr. Yusuf Dilgin, Çanakkale Onsekiz Mart University, Turkey
The seminar will be held in Aula V, Caglioti Building (CU032), on 28 september at 4 pm.
Abstract
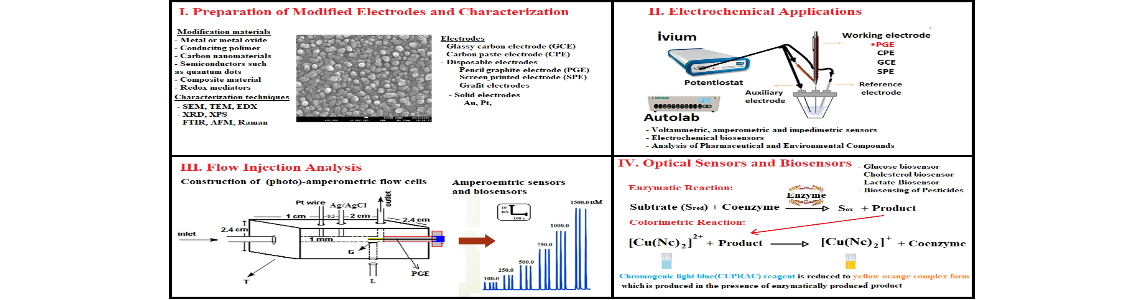
Modification of electrodes is a very useful procedure used in electroanalytical chemistry because modified electrodes exhibit a remarkable improvement in the sensitivity, selectivity, stability, precision, and accuracy of sensors or biosensors compared to unmodified electrodes (1). Therefore, chemically modified electrodes (CMEs) have gained great attention in electrochemical studies in the last two decades. CMEs can be prepared from various materials, such as conducting polymers, metal nanoparticles, carbon nanotubes, bio-recognition elements, quantum dots, and graphene oxide, using different modification strategies. The choice of the electrode material can also affect the cost, selectivity, sensitivity, and stability of an electrochemical sensor/biosensor. Recently, chemically modified disposable electrodes such as pencil graphite electrode (PGE), indium tin oxide (ITO) electrodes, and screen printed electrodes (SPEs) have found great attention in electrochemical studies due to their advantages such as their high electrochemical reactivity, commercial availability, good mechanical rigidity, lower cost, and ease of modification (2-4). Among the disposable electrodes, PGEs have recently found great attention in electroanalytical studies due to their extremely low price, high sensitivity, and ease of commercial availability. Pencil leads are made of graphite, clay, and wax, and their percentages can vary depending on the hardness of the leads. Although bare or pretreated PGEs have been widely used in electrochemical sensor/biosensor studies, these electrodes generally offer poor sensitivity and selective results for the detection of many analytes due to their low electrocatalytic activities. In order to obtain more sensitive and selective results, the PGE surface has been modified with a suitable electrocatalyst or electron mediator in the fabrication of electrochemical sensors/biosensors. In our studies, the determination of a wide range of different environmental and pharmaceutical substances, such as pesticides, heavy metals, drugs used in the treatment of COVID-19, and several biological compounds, has been performed by using electrochemical techniques in different modes, such as voltammetry, amperometry integrated with flow injection analysis (FIA), and electrochemical impedance spectroscopy. In this context, modified PGEs have been used in flow injection analysis (FIA) systems for the first time. It has been concluded that the modification of electrodes significantly increases the capability and sensitivity of electrochemical sensors and biosensors due to their large active surface area, good electrical conductivity, and high surface/volume ratio. It has performed voltammetric determination of many electroactive environmental and pharmaceutical compounds at very low limits, especially with the use of disposable pencil graphite electrodes, which offer several advantages over other carbon-based electrodes. In addition, the modified and unmodified electrodes used in our studies were integrated into the FIA system, resulting in a much more sensitive, simple, and fast analysis of various environmental and pharmaceutical compounds.
Keywords: Electrochemical sensors and biosensors; modified electrodes; flow injection analysis; disposable electrodes.
References
1. Chillawar, R.R.; Tadi, K.K.; Motghare, R.V., Voltammetric Techniques at Chemically Modified Electrodes,
J. Anal. Chem. 2015, 70, 399–418.
2. David I.G.; Buleandra M.; Popa D.E.; Cheregi M.C.; David V.; Iorgulescu E.E.; Tartareanu G.O. Recent developments in voltammetric analysis of pharmaceuticals using disposable pencil graphite electrodes, Processes, 2022, 10, 472.
3. Uygun, Z.O.; Dilgin, Y., A novel impedimetric sensor based on molecularly imprinted polypyrrole modified pencil graphite electrode for trace level determination of chlorpyrifos, Sens. Actuators B., 2013, 188, 78-84.
4. Ayaz, S.; Dilgin, Y.; Apak, R., Flow injection amperometric determination of hydrazine at a cupricneocuproine complex/anionic surfactant modified disposable electrode, Microcjem J., 2020, 159, 105457.
